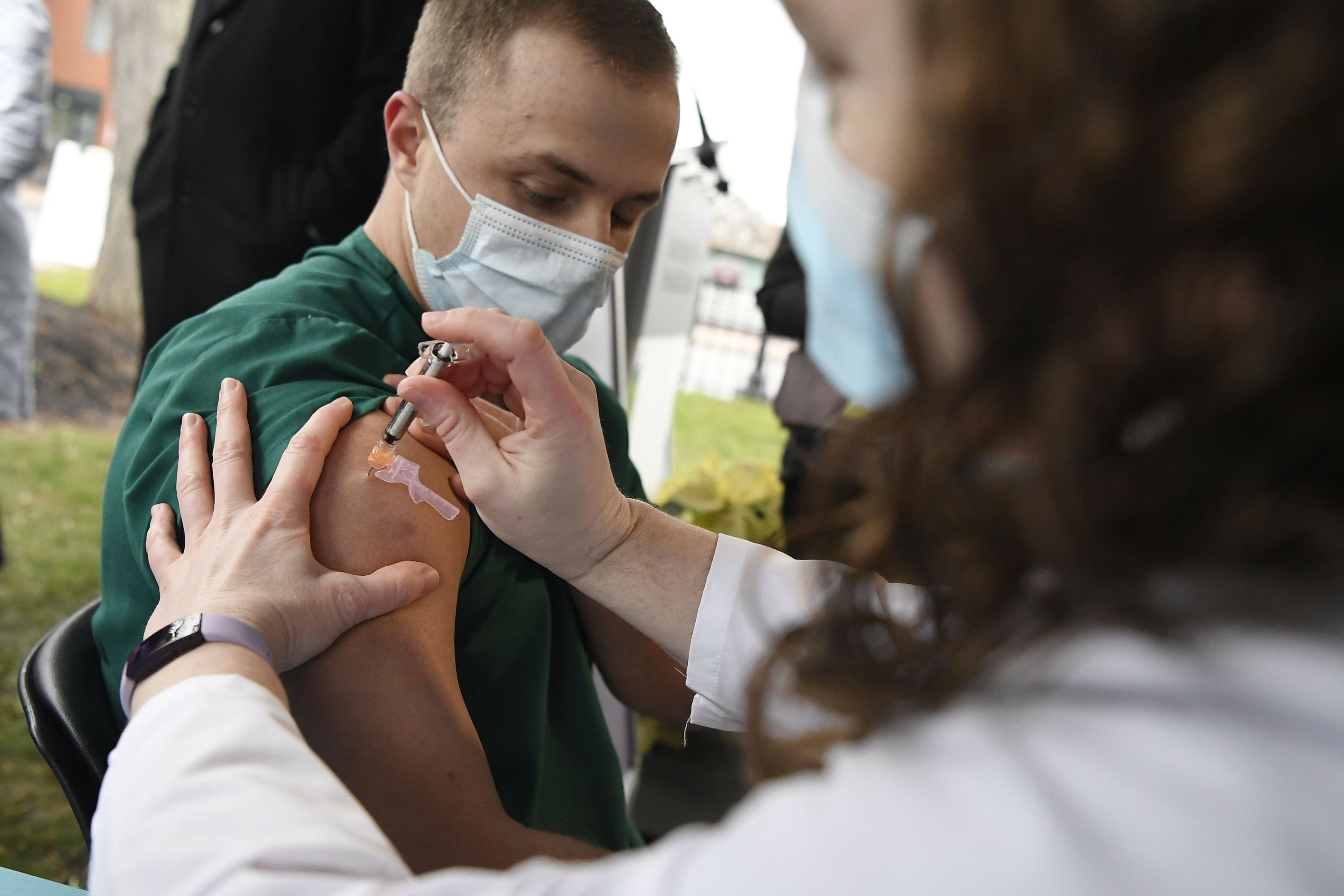
WASHINGTON (AP) – Hundreds of US hospitals will begin vaccinating their employees Tuesday as federal health officials review a second COVID-19 shot needed to spur the nation’s largest vaccination campaign.
Wrapped in dry ice to stay at ultra-frozen temperatures, shipments of Pfizer’s COVID-19 vaccine to 400 additional hospitals and other distribution locations will arrive, a day after the country’s death toll surpassed a whopping 300,000. The first 3 million shots are strictly rationed to primary care workers and elderly care patients, with hundreds of millions of additional injections needed in the coming months to protect most Americans.
The Food and Drug Administration will publish its analysis of a second thoroughly studied COVID-19 vaccine, which could soon join Pfizer-BioNTech’s in the fight against the pandemic. If FDA advisers give it a positive recommendation on Thursday, the agency may give the green light to drug manufacturer Moderna’s vaccine later this week.
A second vaccine cannot come soon enough, as the country’s daily death toll remains above 2,400 amid more than 210,000 new daily cases, based on weekly averages from data collected by Johns Hopkins University. The devastating toll is only expected to increase in the coming weeks, fueled by leisure travel, family reunions and lax compliance with basic public health measures.


The first vaccine deliveries have given some degree of encouragement to exhausted doctors, nurses and hospital workers across the country.
Johnnie Peoples, a 43-year-old survival flight nurse, was excited and a bit nervous Monday afternoon when he unzipped his flight suit and extended his left arm to become the first person to receive the vaccine at the University of Michigan Medical Center in Ann Arbor.
“Just to be a part of it is a good feeling,” he said.
Since March, he has been transporting seriously ill COVID-19 patients by air from smaller hospitals in the state to the university medical center. It is close and personal work that requires him to adjust the ventilator settings and administer infusions to prevent a significant drop in blood pressure.
In Florida, government officials expect to have 100,000 doses of the vaccine in five state hospitals by Tuesday.
“This is 20,000 doses of hope,” said John Couris, president and chief executive officer of Tampa General Hospital, following Monday’s delivery of 3,900 vaccine vials. Each vial has five doses.
Because the vaccine requires two rounds, the people getting injections now will need a second injection in about three weeks.
The vaccinations would also start on Tuesday in New Jersey, which will distribute approximately 76,000 doses among health workers and nursing home residents. The federal government coordinates mass deliveries by private shipping and distribution companies based on locations chosen by the state’s governors.
After another initial batch of deliveries Wednesday, officials from the Trump administration’s Operation Warp Speed in Washington said they will move 580 additional shipments this weekend.
“We’ll start our drumbeat of continuous implementation of the vaccine as soon as it’s available,” Army General Gustave Perna, chief operating officer for Warp Speed, told reporters Monday. “We package and deliver. It’s a constant stream of available vaccines. “
Admissions for nursing home residents will not begin in most states until next Monday, when some 1,100 institutions will begin vaccinations.
Perna and other US officials reiterated their forecast that 20 million Americans will be able to get their first shots by the end of December, and another 30 million in January.
That projection presupposes swift approval of the vaccine under review this week, co-developed by Moderna and the National Institutes of Health. Like the Pfizer BioNTech vaccine, Moderna’s requires two injections for complete protection.
Last month, Moderna and NIH reported that their shot was found to be nearly 95% effective for different ages and racial groups, according to the results of an ongoing study involving 30,000 people. The main side effects were fatigue, muscle pain and pain at the injection site after the second dose. Those flu-like reactions are common with many vaccines and are a sign that the vaccine is triggering the immune system to fight the virus.
Moderna did not report any major safety concerns from its research. But the FDA panel will certainly examine the data for evidence of possible serious allergic reactions or other rare side effects. Officials in the UK there are investigating several side effects with Pfizer’s vaccine and the FDA is closely following the rollout here for similar reports.
The shots from both Moderna and Pfizer-BioNTech are so-called mRNA vaccines, a brand new technology. They are not made with the coronavirus itself, which means that no one can see it from the photos. Instead, the vaccine contains a piece of genetic code that trains the immune system to recognize the enriched protein on the surface of the virus.
___
Associated Press authors Lindsey Tanner, Tamara Lush, Candice Choi and Lauran Neergaard contributed to this report.
___
The Associated Press Department of Health and Science is supported by the Science Education Department of the Howard Hughes Medical Institute. The AP is solely responsible for all content.
I’m really impressed along with your writing abilities as smartly as with the format on your weblog. Is that this a paid subject matter or did you modify it yourself? Anyway keep up the nice high quality writing, it is rare to look a great weblog like this one these days!