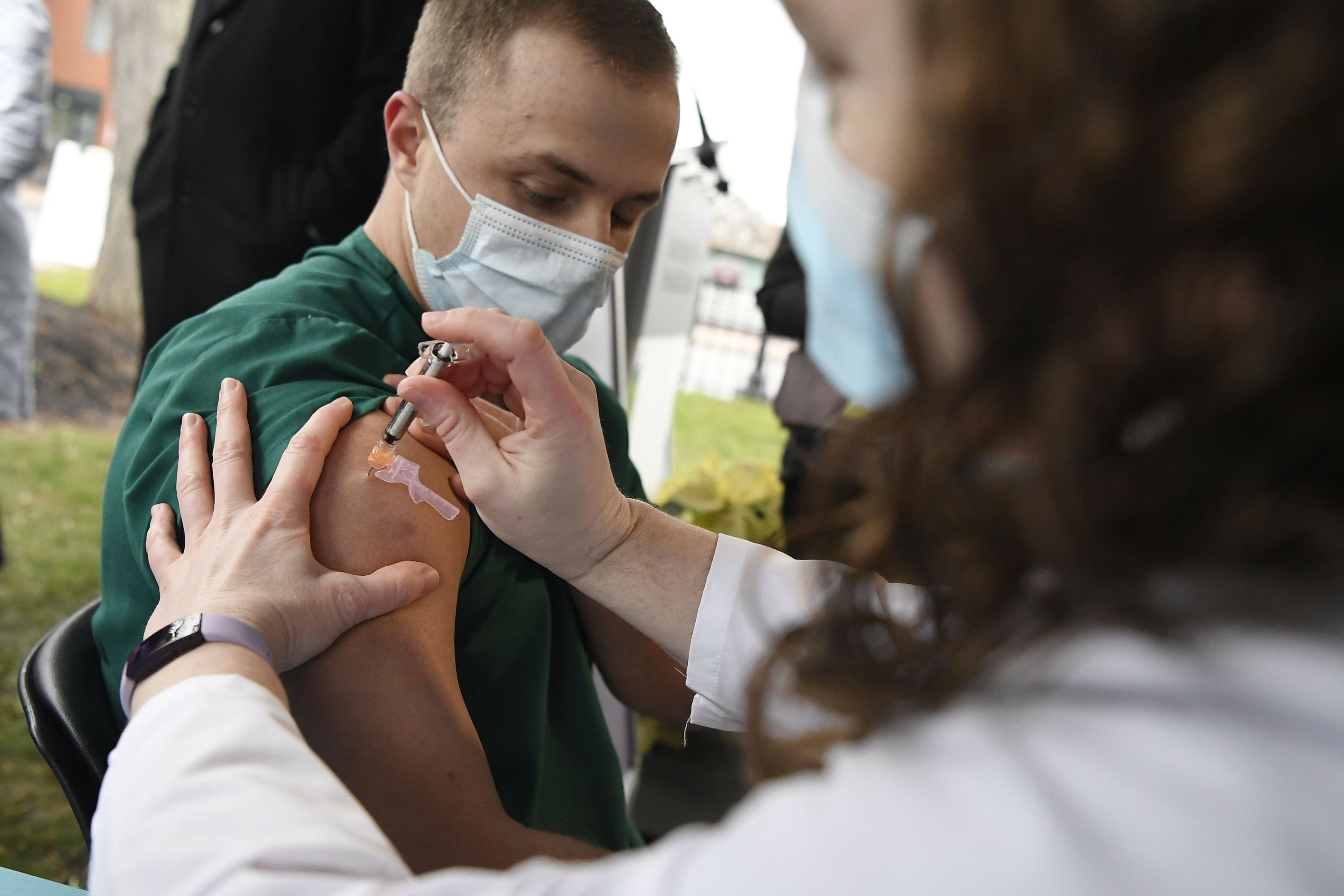
WASHINGTON (AP) – Hundreds of hospitals across the country began providing COVID-19 injections to their employees on Tuesday in a rapid extension of the US round of vaccinations, while a second vaccine was on the cusp of government licensing.
A day after the rollout of Pfizer-BioNTech’s coronavirus shots, the Food and Drug Administration said its preliminary analysis confirmed the effectiveness and safety of the vaccine developed by Moderna and the National Institutes of Health. A panel of outside experts is expected to vote Thursday to recommend the formula, with the FDA’s green light shortly thereafter.
The Moderna vaccine uses the same technology as that of Pfizer-BioNTech and showed similarly strong protection against COVID-19, but is easier to handle because it does not need to be stored in the freezer at minus 94 degrees Fahrenheit (minus 70 degrees Celsius).
Another weapon against the outbreak can’t come soon enough: The US death toll surpassed a whopping 300,000 Monday, according to Johns Hopkins University, with an average of about 2,400 people dying per day and new cases averaging more than 210,000 per day. .
The devastating toll is only expected to increase in the coming weeks, fueled by leisure travel, family reunions and lax compliance with basic public health measures.


Wrapped in dry ice, shipments of the Pfizer-BioNTech vaccine began arriving at 400 additional hospitals and other distribution locations on Tuesday.
The first 3 million shots are strictly rationed to primary care health workers and nursing home patients, and hundreds of millions of shots will be needed over the next few months to protect most Americans.
The rollout has been an encouragement to exhausted doctors, nurses and hospital staff across the country.
Maritza Beniquez was in the front row after the devastation the COVID-19 pandemic has wreaked on communities of color in New Jersey, so she seized the opportunity to take the vaccine hailed as a turning point in the long and grueling battle against the virus.
The 56-year-old nurse in the emergency room at Newark’s University Hospital on Tuesday became the first person in New Jersey to receive the vaccine. All recipients will receive a second admission a few weeks later.
“I am happy that in a month and a half I will no longer have to worry about entering a room. I don’t have to be afraid to perform chest compressions or be present if they intubate a patient, ”said Beniquez. “I don’t want to be afraid anymore, and I don’t want to risk taking it home with my own family and my own friends.”
The federal government plans hundreds of additional shipments over the weekend.
“We will begin our ongoing implementation of the vaccine as soon as it becomes available,” said Army General Gustave Perna, chief operating officer of the government’s Operation Warp Speed. “We package and deliver. It’s a constant stream of available vaccines. “
Shots to nursing home residents won’t begin in most states until next week, when about 1,100 facilities will begin vaccinations. Perna and other officials reiterated their forecast that 20 million Americans will be able to get their first shots by the end of December, and 30 million more by January.
That projection assumes rapid approval of the Moderna vaccine, which also requires two injections for full protection. The US government has purchased 100 million doses of the Pfizer BioNTech vaccine and is ordering 200 million doses of the Moderna serum. Assuming there are no production or distribution delays, that would be enough to vaccinate 150 million Americans by mid-2021.
Elsewhere in the world, the Pfizer BioNTech vaccine is given in Great Britain and Canada. And European Union regulators have moved a meeting to review the vaccine to December 21, more than a week ahead of schedule, amid pressure from Germany and other countries on the continent.
In examining the initial results of a study involving 30,000 individuals, the FDA found that Moderna’s vaccine worked much the same as that of Pfizer-BioNTech.
The Moderna vaccine was more than 94% effective in preventing COVID-19 disease overall, and 86% effective in people aged 65 and older. The FDA has not identified any major safety concerns. Side effects can include fever, fatigue, and pain as the vaccine activates the immune system.
Even such a large study cannot detect very rare problems. But the FDA looked carefully for signs of allergic reactions after Britain reported some possible reactions last week in people with a history of severe allergies who received the Pfizer-BioNTech injection.
The FDA found no serious allergic reactions in the Moderna study. About 1.5% of vaccine recipients and 1.1% who received sham injections reported potentially minor “hypersensitivity reactions”.
The shots from both Moderna and Pfizer-BioNTech are so-called mRNA vaccines. They are not made with the coronavirus itself, which means that there is no chance that anyone will be able to see it from the photos.
Instead, the vaccine contains a piece of genetic code that trains the immune system to recognize the enriched protein on the surface of the virus.
___
Associated Press writers Tamara Lush and Candice Choi contributed to this report.
___
The Associated Press Department of Health and Science is supported by the Science Education Department of the Howard Hughes Medical Institute. The AP is solely responsible for all content.